Serum Klotho as a feasible diagnostic biomarker for metabolic syndrome in Iraqi adults aged over 50 years
Main Article Content
Abstract
Background: Metabolic Syndrome (MetS) is a cluster of cardiometabolic risk factors primarily driven by insulin resistance, with a high prevalence in older adult populations.
Objectives: This study aimed to evaluate the diagnostic accuracy of serum Klotho Beta (KLB) and insulin as novel biomarkers for MetS in Iraqi adults over 50 years old.
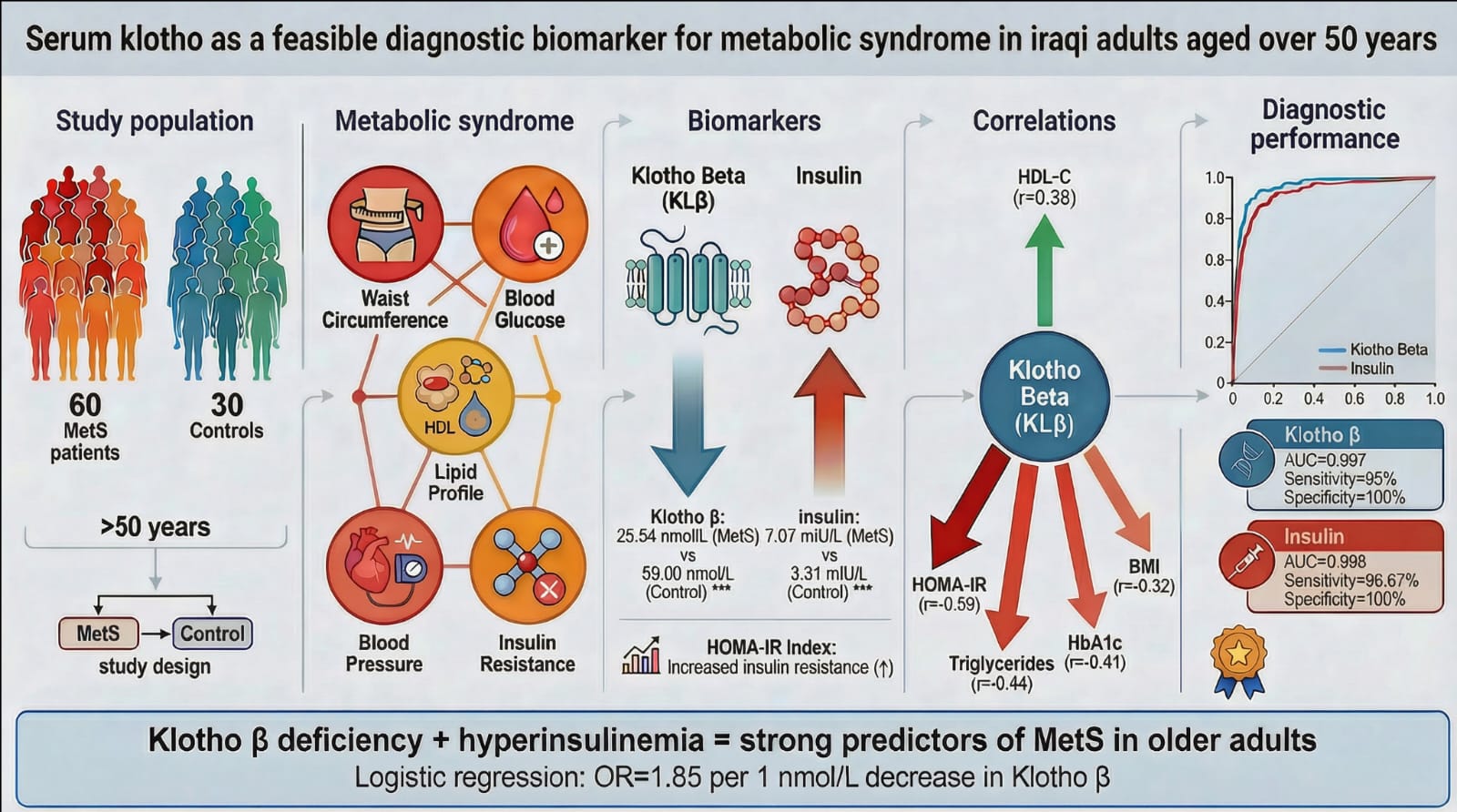
Materials and methods: This case-control study included 60 MetS patients and 30 healthy controls, recruited from Iraqi hospitals. MetS was diagnosed using harmonized IDF/AHA/NCEP ATP III criteria. Serum Klotho Beta and insulin levels were measured by ELISA.
Results: Serum Klotho Beta levels were significantly lower in the MetS group (25.54±4.41 nmol/L) compared to controls (59.00±11.05 nmol/L, p<0.001). Klotho Beta was inversely correlated with HOMA-IR (r=-0.59, p<0.001), BMI, HbA1c, and triglycerides, and positively with HDL-C. ROC analysis showed an AUC of 0.997 for Klotho Beta (sensitivity 95%, specificity 100% at ≤35.248 nmol/L) and an AUC of 0.998 for insulin. Logistic regression confirmed Klotho Beta as a strong independent predictor of MetS (OR=1.85 per 1 nmol/L decrease).
Conclusion: Serum Klotho Beta and insulin are highly sensitive and specific biomarkers for MetS in older adults. Klotho Beta deficiency, coupled with hyperinsulinemia, reflects underlying insulin resistance and metabolic dysregulation, highlighting its potential for risk stratification and early diagnosis in high-risk populations.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Personal views expressed by the contributors in their articles are not necessarily those of the Journal of Associated Medical Sciences, Faculty of Associated Medical Sciences, Chiang Mai University.
References
Chew NWS, Ng CH, Tan DJH, et al. The global burden of metabolic disease: Data from 2000 to 2019. Cell Metab. 2023; 35(3): 414-28.e3. doi:10.1016/j.cmet.2023.02.003.
Neeland IJ, Lim S, Tchernof A, et al. Metabolic syndrome. Nat Rev Dis Prim. 2024; 10(1): 60-77. doi:10.1038/s41572-024-00563-5.
Nwankwo M, Okamkpa CJ, Danborno B. Comparison of diagnostic criteria and prevalence of metabolic syndrome using WHO, NCEP-ATP III, IDF and harmonized criteria: A case study from urban southeast Nigeria. Diabetes Metab Syndr Clin Res Rev. 2022; 16(12): 1-34. doi: 10.1016/j.dsx.2022.102665.
Fahed G, Aoun L, Bou Zerdan M, et al. Metabolic syndrome: updates on pathophysiology and management in 2021. Int J Mol Sci. 2022; 23(2): 786. doi: 10.3390/ijms23020786.
Liu L, Cai XC, Sun XY, et al. Global prevalence of metabolic syndrome in patients with psoriasis in the past two decades: current evidence. J Eur Acad Dermatol Venereol. 2022; 36(11): 1969-79. doi:10.1111/jdv.18296.
Jamali Z, Ayoobi F, Jalali Z, et al. Metabolic syndrome: a population-based study of prevalence and risk factors. Sci Rep. 2024; 14(1): 3987. doi:10.1038/s41598-024-54367-4.
Al-Rubeaan K, Bawazeer N, Al Farsi Y, et al. Prevalence of metabolic syndrome in Saudi Arabia-a cross sectional study. BMC Endocr Disord. 2018; 18(1): 1-9. doi: 10.1186/s12902-018-0244-4.
Liang X, Or B, Tsoi MF, Cheung CL, Cheung BMY. Prevalence of metabolic syndrome in the United States National Health and Nutrition Examination Survey 2011-18. Postgrad Med J. 2023; 99(1175): 985-92. doi:10.1093/postmj/qgad008.
Pigeot I, Ahrens W. Epidemiology of metabolic syndrome. Pflugers Arch. 2025; 477(5): 669-80. doi:10.1007/s00424-024-03051-7.
Bowo-Ngandji A, Kenmoe S, Ebogo-Belobo JT, et al. Prevalence of the metabolic syndrome in African populations: A systematic review and meta-analysis. PLoS One. 2023; 18(7): e0289155. doi:10.1371/journal.pone.0289155.
Noubiap JJ, Nansseu JR, Lontchi-Yimagou E, et al. Geographic distribution of metabolic syndrome and its components in the general adult population: A meta-analysis of global data from 28 million individuals. Diabetes Res Clin Pract. 2022; 188: 109924. doi: 10.1016/j.diabres.2022.109924.
Srikanthan K, Feyh A, Visweshwar H, Shapiro JI, Sodhi K. Systematic review of metabolic syndrome biomarkers: A panel for early detection, management, and risk stratification in the West Virginian population. Int J Med Sci. 2016; 13(1): 25-38. doi:10.7150/ijms.13800.
Warner JD, Blake GM, Garrett JW, et al. Correlation of HbA1c levels with CT-based body composition biomarkers in diabetes mellitus and metabolic syndrome. Sci Rep. 2024; 14(1): 21875. doi:10.1038/s41598-024-72702-7.
Geng L, Yi X, Lin Y, et al. Site-specific analysis and functional characterization of N-linked glycosylation for β-Klotho protein. Int J Biol Macromol. 2025; 289: 138846. doi: 10.1016/j.ijbiomac.2024.138846.
Yang M, Liu C, Jiang N, et al. Fibroblast growth factor 21 in metabolic syndrome. Front Endocrinol (Lausanne). 2023; 14: 1220426. doi: 10.3389/fendo.2023.1220426.
Wu SE, Chen WL. Soluble klotho as an effective biomarker to characterize inflammatory states. Ann Med. 2022; 54(1): 1520-9. doi:10.1080/07853890.2022.2077428.
Zhou J, Ben S, Xu T, Xu L, Yao X. Serum β-klotho is a potential biomarker in the prediction of clinical outcomes among patients with NSCLC. J Thorac Dis. 2021; 13(5): 3137-50. doi:10.21037/jtd-21-798.
ElSayed NA, Aleppo G, Aroda VR, et al. Introduction and methodology: standards of care in diabetes-2023. Diabetes Care. 2023; 46(Suppl_1): S1-S4. doi: 10.2337/dc23-Sint.
Li S, Wang P, Wang J, Zhao J, Wang X, Liu T. Effect of mind-body exercise on risk factors for metabolic syndrome including insulin resistance: a metaanalysis. Front Endocrinol (Lausanne). 2024; 15: 1289254. doi: 10.3389/fendo.2024.1289254.
Yuguang L, Chang Y, Chen N, et al. Serum klotho as a novel biomarker for metabolic syndrome: findings from a large national cohort. Front Endocrinol (Lausanne). 2024; 15: 1-8. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2024.1295927.
Kuro-o M. The Klotho proteins in health and disease. Nat Rev Nephrol. 2019; 15(1): 27-44. doi: 10.1038/s41581-018-0078-3.
Liang Y, Liu Y, Tan Q, Zhou K, Wu Y, Yu L. Systemic immune-inflammation mediates the association between Klotho protein and metabolic syndrome: findings from a large-scale population-based study. Lipids Health Dis. 2024; 23(1): 360. doi:10. 1186/s12944-024-02339-y.
Lin Y, Chen J, Sun Z. Antiaging gene Klotho deficiency promoted high-fat diet–induced arterial stiffening via inactivation of AMP-activated protein kinase. Hypertension. 2016; 67(3): 564-73. doi: 10.1161/HYPERTENSIONAHA.115.06825.
Hu MC, Shi M, Zhang J, et al. Klotho deficiency causes vascular calcification in chronic kidney disease. J Am Soc Nephrol. 2011; 22(1): 124-36. doi: 10.1681/ASN.2009121311%0A.
Jiang S, Wang Y, Wang Z, Zhang L, Jin F, Li B. The association of serum Klotho concentrations with hyperlipidemia prevalence and lipid levels among US adults: a cross-sectional study. BMC Public Health. 2023; 23(1): 1-12. doi: 10.1186/s12889-023-16566-y.
Keuper M, Häring HU, Staiger H. Circulating FGF21 levels in human health and metabolic disease. Exp Clin Endocrinol Diabetes. 2020; 128(11): 752-70. doi: 10.1055/a-0879-2968.
Bagchi DP, Li Z, Corsa CA, et al. Wntless regulates lipogenic gene expression in adipocytes and protects against diet-induced metabolic dysfunction. Mol Metab. 2020; 39: 100992. doi: 10.1016/j.molmet.2020.100992.
Wyble K. Implementing the American Diabetes Association Glycemic Assessment Clinical Practice Guidelines in the care of patients diagnosed with type 2 diabetes in a single rural healthcare clinic in Southwest Louisiana. University of Louisiana at Lafayette; 2023.
Tsai SF, Yang CT, Liu WJ, Lee CL. Development and validation of an insulin resistance model for a population without diabetes mellitus and its clinical implication: a prospective cohort study. EClinicalMedicine. 2023; 58: 101934. doi:10. 1016/j.eclinm.2023.101934.
Kawada T. Insulin-related biomarkers to predict the risk of metabolic syndrome. Int J Endocrinol Metab. 2013; 11(4): e10418. doi:10.5812/ijem.10418.
Gao K, Lin Y. Insulin resistance mediates the association between physical activity and mortality in US adults with metabolic syndrome. Sci Rep. 2025; 15(1): 18872. doi:10.1038/s41598-025-02921-z
Nolan CJ, Prentki M. Insulin resistance and insulin hypersecretion in the metabolic syndrome and type 2 diabetes: Time for a conceptual framework shift. Diabetes Vasc Dis Res. 2019; 16(2): 118-27. doi: 10.1177/1479164119827611.
Kim S, Song K, Lee M, et al. Trends in HOMA-IR values among South Korean adolescents from 2007–2010 to 2019–2020: a sex-, age-, and weight status-specific analysis. Int J Obes. 2023; 47(9): 865-72. doi: 10.1177/1479164119827611.
Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev. 2018; 98(4).1-91 doi: 10.1152/physrev.00063.2017.
Meroni M, Dongiovanni P, Tiano F, Piciotti R, Alisi A, Panera N. β-Klotho as novel therapeutic target in Metabolic dysfunction-associated steatotic liver disease (MASLD): A narrative review. Biomed Pharmacother. 2024; 180: 117608. doi: 10.1016/j.biopha.2024.117608.
Islam MS, Wei P, Suzauddula M, et al. The interplay of factors in metabolic syndrome: understanding its roots and complexity. Mol Med. 2024; 30(1): 1-21. doi:10.1186/s10020-024-01019-y.
Bovolini A, Garcia J, Andrade MA, Duarte JA. Metabolic syndrome pathophysiology and predisposing Factors. Int J Sports Med. 2021;42(3): 199-214. doi:10.1055/a-1263-0898.
Hooshmand E, Akbarzade I, Delbari D, Niroomand M, Ghavidel F, Saatchi M. Prevalence of metabolic syndrome among adults aged 50 and above and associated factors: A cross-sectional study from Ardakan cohort study on aging (ACSA). Heal Sci Reports. 2025; 8(3):1-7. doi: 10.1002/hsr2.70508.
Holmannova D, Borsky P, Andrys C, et al. The influence of metabolic syndrome on potential aging biomarkers in participants with metabolic syndrome compared to healthy controls. Biomedicines. 2024; 12(1): 1-15. doi:10.3390/biomedicines12010242.