Brief review: Effects of textural properties of lamellar silica in drug delivery applications
Main Article Content
Abstract
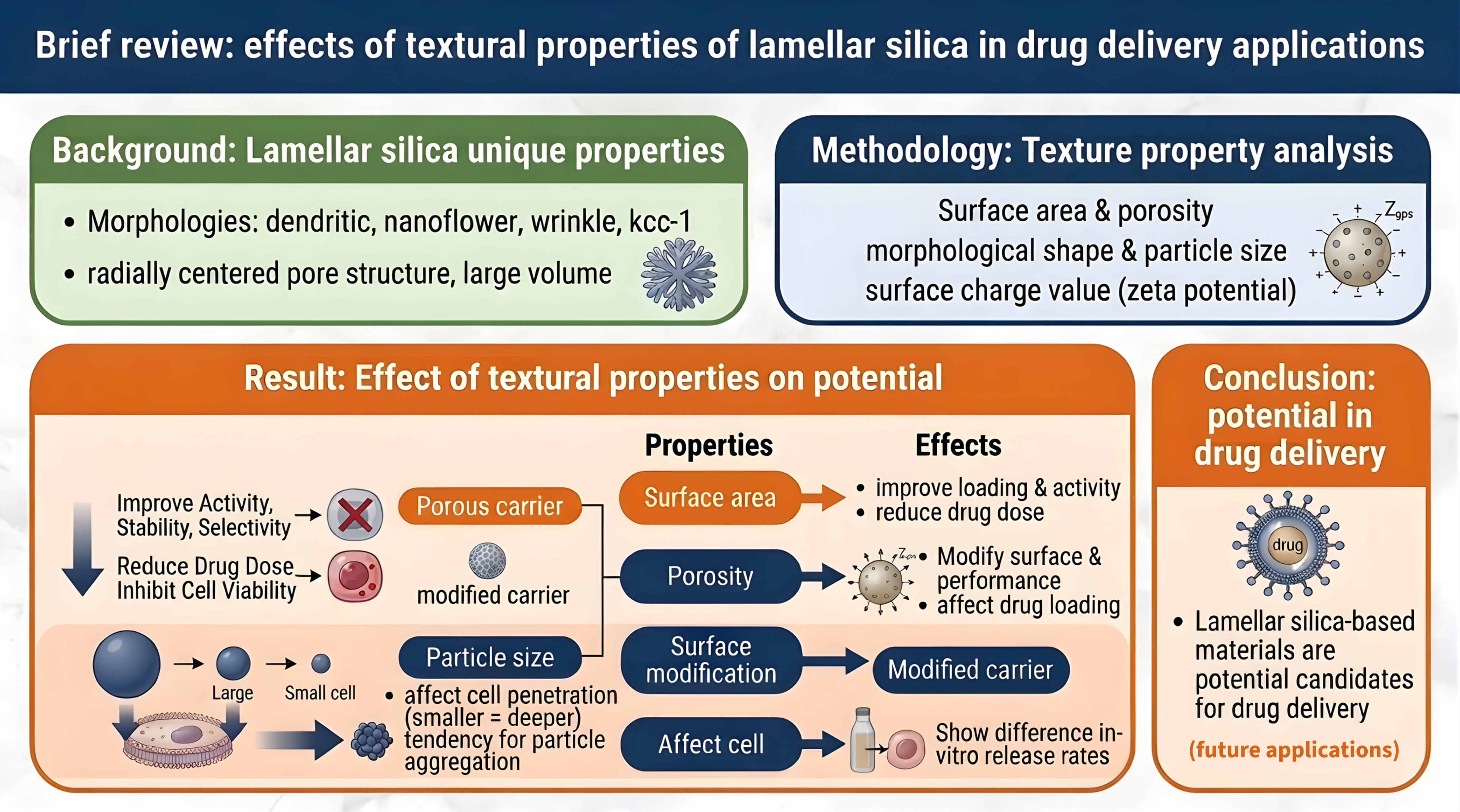
Background: Silica-based materials, especially those with lamellar morphology, usually have other names, such as dendritic, nanoflower, and wrinkle are often used in various applications due to their unique textural properties, such as radially centered pore structure, pore channels, and large pore volume.
Objectives: To determine the effect of lamellar silica texture properties, such as surface area, porosity, pore size, morphological shape, particle size, and surface charge value, as potential drug delivery material candidates. Materials and methods: The texture properties of silica materials were reviewed in drug delivery applications, and four silica materials with different morphologies were reviewed for their texture properties and potential in drug delivery applications.
Results: The literature review revealed the effect of material texture properties on their potential in drug delivery applications. Drug loading in porous carriers can improve their activity, stability, and selectivity, and can reduce the drug dose to inhibit cell viability compared to unencapsulated drugs. Surface modification can also affect the zeta potential value and its performance in drug loading. In vitro drug release properties of the material and drug release rates showed different results related to their different texture properties. Particle size affects particle penetration into cells; the smaller the particle size, will cause deeper the penetration, but it has a tendency for particle aggregation.
Conclusion: Lamellar silica-based materials with various unique properties of their texture properties show great potential in drug delivery applications and are expected to be used in the future.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Personal views expressed by the contributors in their articles are not necessarily those of the Journal of Associated Medical Sciences, Faculty of Associated Medical Sciences, Chiang Mai University.
References
Esmaeili Y, Dabiri A, Mashayekhi F, Rahimmanesh I, Bidram E, Karbasi S, et al. Smart co-delivery of plasmid DNA and doxorubicin using MCMchitosan-PEG polymerization functionalized with MUC-1 aptamer against breast cancer. Biomed Pharmacother. 2024; 173: 116465. doi: 10.1016/j.biopha.2024.116465.
Septian Dwitya S, Lin K-S, Weng M-T, Vukile Mdlovu N, Yang M-T, Wu C-M. Synthesis and characterization of oleic acid-stabilized cobalt ferrite @MCM-41/nanocomposites for pH-responsive drug delivery. J Ind Eng Chem. 2024; doi: 10.1016/j.jiec.2024.08.036.
Patel A, Mehta S. Functionalized SBA-15: engineering, detailed study on release and kinetics of alendronate as well as its anti-tumour properties towards osteosarcoma. RSC Pharm. 2024; 1(4): 797– 805. doi: 10.1039/D4PM00078A.
Atiyah NA, Albayati TM, Atiya MA. Interaction behavior of curcumin encapsulated onto functionalized SBA-15 as an efficient carrier and release in drug delivery. J Mol Struct. 2022; 1260: 132879. doi: 10.1016/j.molstruc.2022.132879.
Nguyen H-VT, Mai NXD, Thi Lai H, Nguyen LHT, Pham QT, Van Nguyen H, et al. Targeted, pH responsive quercetin delivery via hyaluronic acid-functionalized mesoporous nanoparticles for cancer treatment. Colloids Surfaces A Physicochem Eng Asp. 2024; 135914. doi: 10.1016/j.colsurfa. 2024. 135914.
Lestari WA, Saputra OA, Díaz-García D, Wahyuningsih S, Gómez-Ruiz S, Wibowo FR. Design of experiments as a strategy for modulating the colloidal stability, physico-chemical properties and drugdelivery potential of small mesoporous silica nanoparticles. Adv Powder Technol. 2023; 34(10): 104191. doi: 10.1016/j.apt.2023.104191.
Yang J, Liu B, Wang Q, Yan H, Li G, Wang X, et al. Carboxylated mesoporous silica nanoparticlenucleic acid chimera conjugate-assisted delivery of siRNA and doxorubicin effectively treat drug-resistant bladder cancer. Biomed Pharmacother. 2024; 178: 117185. doi: 10.1016/j.biopha.2024.117185.
Polshettiwar V. Dendritic Fibrous Nanosilica: Discovery, synthesis, formation mechanism, catalysis, and CO2 capture-conversion. Acc Chem Res. 2022; 55(10): 1395-410. doi: 10.1021/acs.accounts.2c00031.
Wang Y, Nor YA, Song H, Yang Y, Xu C, Yu M, et al. Small-sized and large-pore dendritic mesoporous silica nanoparticles enhance antimicrobial enzyme delivery. J Mater Chem B. 2016; 4(15): 2646-53. doi: 10.1039/C6TB00053C.
Shen D, Yang J, Li X, Zhou L, Zhang R, Li W, et al. Biphase stratification approach to threedimensional dendritic biodegradable mesoporous silica nanospheres. Nano Lett. 2014; 14(2): 923-32. doi: 10.1021/nl404316v.
Yang X, Quan K, Wang J, Liu J, Liu B, Chen J, et al. Particle size and pore adjustment of dendritic mesoporous silica using different long alkylchain imidazolium ionic liquids as templates. Microporous Mesoporous Mater. 2022; 345: 112249. doi: 10.1016/j.micromeso.2022.112249.
Sun Z, Cui G, Li H, Tian Y, Yan S. Multifunctional dendritic mesoporous silica nanospheres loaded with silver nanoparticles as a highly active and recyclable heterogeneous catalyst. Colloids Surfaces A Physicochem Eng Asp. 2016; 489: 142-53. doi: 10.1016/j.colsurfa.2015.10.052.
Hao P, Peng B, Shan B-Q, Yang T-Q, Zhang K. Comprehensive understanding of the synthesis and formation mechanism of dendritic mesoporous silica nanospheres. Nanoscale Adv. 2020; 2(5): 1792- 810. doi: 10.1039/D0NA00219D.
Maity A, Polshettiwar V. Dendritic fibrous nanosilica for catalysis, energy harvesting, carbon dioxide mitigation, drug delivery, and sensing. ChemSusChem. 2017; 10(20): 3866-913. doi: 10.1002/cssc.201701076.
Cocuzza C, Vincenzi C, Ottone C, Illanes A, Fino D, Cauda V, et al. Synthesis and characterization of mesoporous silicas with dendritic and spongy-like structures: Potential supports for human lactate dehydrogenase-based microreactors aimed at anticancer inhibitor screening. MicroporousMesoporous Mater. 2024; 376: 113182. doi: 10.1016/j.micromeso.2024.113182.
Li X, Yang S, Luan Y, Wang D, Du X. Polydopamine modification on dendritic porous silica surface for efficient adhesion of functional nanoparticles. Colloids Surfaces A Physicochem Eng Asp. 2024; 703: 135439. doi: 10.1016/j.colsurfa. 2024.135439.
Ovejero-Paredes K, Díaz-García D, Mena-Palomo I, Marciello M, Lozano-Chamizo L, Morato YL, et al. Synthesis of a theranostic platform based on fibrous silica nanoparticles for the enhanced treatment of triple-negative breast cancer promoted by a combination of chemotherapeutic agents. Biomater Adv. 2022; 137: 212823. doi: 10.1016/j.bioadv.2022.212823.
Febriyanti E, Suendo V, Mukti RR, Prasetyo A, Arifin AF, Akbar MA, et al. Further insight into the definite morphology and formation mechanism of mesoporous silica KCC-1. Langmuir. 2016; 32(23): 5802-11. doi: 10.1021/acs.langmuir.6b00675.
Pérez-Moreno AM, Aranda CJ, Torres MJ, Mayorga C, Paris JL. Immunomodulatory potential of rapamycin-loaded mesoporous silica nanoparticles: pore size-dependent drug loading, release, and in vitro cellular responses. Drug Deliv Transl Res. 2024; 14(12): 3467-76. doi: 10.1007/s13346-02401575-0.
Farzan M, Roth R, Schoelkopf J, Huwyler J, Puchkov M. The processes behind drug loading and release in porous drug delivery systems. Eur J Pharm Biopharm. 2023; 189: 133-51. doi: 10.1016/j.ejpb.2023.05.019.
Ly P-D, Ly K-N, Phan H-L, Nguyen HHT, Duong V-A, Nguyen H V. Recent advances in surface decoration of nanoparticles in drug delivery. Front Nanotechnol. 2024; 6. doi: 10.3389/fnano.2024.1456939.
Mok ZH. The effect of particle size on drug bioavailability in various parts of the body. Pharm Sci Adv. 2024; 2: 100031. doi: 10.1016/j.pscia.2023.100031.
Farahmandghavi F, Imani M, Hajiesmaeelian F. Silicone matrices loaded with levonorgestrel particles: Impact of the particle size on drug release. J Drug Deliv Sci Technol. 2019; 49: 132-42. doi: 10.1016/j.jddst.2018.10.029.
Islam MA, Barua S, Barua D. A multiscale modeling study of particle size effects on the tissue penetration efficacy of drug-delivery nanoparticles. BMC Syst Biol. 2017; 11(1): 113. doi: 10.1186/ s12918-017-0491-4.
Honary S, Zahir F. Effect of zeta potential on the properties of nano-drug delivery systems - A review (Part 1). Trop J Pharm Res. 2013; 12(2). doi: 10.4314/tjpr.v12i2.19.
Feng X, Jia P, Zhang D. Nanocarrier drug delivery system: promising platform for targeted depression therapy. Front Pharmacol. 2024; 15. doi: 10.3389/fphar.2024.1435133.
Naik SP, Elangovan SP, Okubo T, Sokolov I. Morphology control of mesoporous silica particles. J Phys Chem C. 2007; 111(30): 11168-73. doi: 10.1021/jp072184a.
Atluri R, Jensen KA. Engineered nanomaterials: Their physicochemical characteristics and how to measure them. MODENA. 2017: pp3-23. doi: 10.1007/978-3-319-47754-1_1.
Knežević NŽ, Durand J-O. Large pore mesoporous silica nanomaterials for application in delivery of biomolecules. Nanoscale. 2015; 7(6): 2199-209. doi: 10.1039/C4NR06114D.
Polshettiwar V, Cha D, Zhang X, Basset JM. Highsurface-area silica nanospheres (KCC-1) with a fibrous morphology. Angew Chemie Int Ed. 2010; 49(50): 9652-6. doi: 10.1002/anie.201003451.
Maity A, Polshettiwar V. Scalable and sustainable synthesis of size-controlled monodisperse dendritic fibrous nanosilica quantified by E-factor. ACS Appl Nano Mater. 2018; 1(7): 3636-43. doi: 10.1021/acsanm.8b00761.
Megantari V, Febriyanti E, Benu DP, Reza M, Steky F V., Yuliarto B, et al. Nanofluidic behavior of diatomic molecules in bicontinuous concentric lamellar (bcl) silica formed by polysiloxane solgel phase segregation as a reference in the mass transport through the open channel system. Polym Technol Mater. 2020; 59(12): 1359-69. doi: 10.1080/25740881.2020.1738478.
Guo Z, Wu L, Wang Y, Zhu Y, Wan G, Li R, et al. Design of dendritic large-pore mesoporous silica nanoparticles with controlled structure and formation mechanism in dual-templating strategy. ACS Appl Mater Interfaces. 2020; 12(16): 18823-32. doi: 10.1021/acsami.0c00596.
Yin S, Xu C, Yang H, Wu C, Wu M, Xu J, et al. Facilely preparing highly dispersed Ni-based CO2 methanation catalysts via employing the aminofunctionalized KCC-1 support. Fuel. 2024; 365: 131162. doi: 10.1016/j.fuel.2024.131162.
Lai LW, Teh LP, Timmiati SN, Kamarudin NHN, Setiabudi HD. A sustainable solution for diclofenac adsorption: Chitosan-modified fibrous silica KCC-1 adsorbent. J Environ Chem Eng. 2023; 11(6): 111295. doi: 10.1016/j.jece.2023.111295.
Silmi N, Febriyanti E, Andriani A, Arsyad R, Steky FV, Mukti RR, et al. Textural control of bicontinuous concentric lamellar nanostructured mesoporous silica via low-pressure solvothermal method. Mater Chem Phys. 2021; 265: 124492. doi: 10.1016/j.matchemphys.2021.124492.
Kole K, Halder A, Singh S, Samanta A, Das S, Kundu AK, et al. Chromogenic-functionalized silica nanoflower composites for the detection of carbon dioxide. ACS Appl Nano Mater. 2020; 3(5): 4321-8. doi: 10.1021/acsanm.0c00469.
Palanichamy K, Umasankar S, Ganesh S, Sasirekha N. Highly coke resistant Ni–Co/KCC-1 catalysts for dry reforming of methane. Int J Hydrogen Energy. 2023; 48(31): 11727-45. doi: 10.1016/j.ijhydene.2022.12.076.
Thommes M, Kaneko K, Neimark A V., Olivier JP, Rodriguez-Reinoso F, Rouquerol J, et al. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem. 2015; 87(9–10): 1051-69. doi: 10.1515/pac-2014-1117.
Latifi L, Sohrabnezhad S. Influence of pore size and surface area of mesoporous silica materilas (MCM41 and KIT-6) on the drug loading and release. J Sol-Gel Sci Technol. 2018; 87(3): 626-38. doi: 10.1007/s10971-018-4742-7.
Jia L, Shen J, Li Z, Zhang D, Zhang Q, Liu G, et al. In vitro and in vivo evaluation of paclitaxel-loaded mesoporous silica nanoparticles with three pore sizes. Int J Pharm. 2013; 445(1-2): 12-9. doi: 10.1016/j.ijpharm.2013.01.058.
Peng S, Huang B, Lin Y, Pei G, Zhang L. Effect of surface functionalization and pore structure type on the release performance of mesoporous silica nanoparticles. Microporous Mesoporous Mater. 2022; 336: 111862. doi: 10.1016/j.micromeso.2022.111862.
Bavnhøj CG, Knopp MM, Madsen CM, Löbmann K. The role interplay between mesoporous silica pore volume and surface area and their effect on drug loading capacity. Int J Pharm X. 2019; 1: 100008. doi: 10.1016/j.ijpx.2019.100008.
Mujtaba M, Negi A, King AWT, Zare M, Kuncova-Kallio J. Surface modifications of nanocellulose for drug delivery applications; a critical review. Curr Opin Biomed Eng. 2023; 28: 100475. doi: 10.1016/j.cobme.2023.100475.
Wang J, Wang Y, Liu Q, Yang L, Zhu R, Yu C, et al. Rational design of multifunctional dendritic mesoporous silica nanoparticles to load curcumin and enhance efficacy for breast cancer therapy. ACS Appl Mater Interfaces. 2016; 8(40): 26511-23. doi: 10.1021/acsami.6b08400.
Pishnamazi M, Hafizi H, Pishnamazi M, Marjani A, Shirazian S, Walker GM. Controlled release evaluation of paracetamol loaded amine functionalized mesoporous silica KCC1 compared to microcrystalline cellulose based tablets. Sci Rep. 2021; 11(1): 535. doi: 10.1038/s41598-020-79983-8.
Maity A, Das A, Sen D, Mazumder S, Polshettiwar V. Unraveling the formation mechanism of dendritic fibrous nanosilica. Langmuir. 2017; 33(48): 13774-82. doi: 10.1021/acs.langmuir.7b02996.
Deng C, Liu Y, Zhou F, Wu M, Zhang Q, Yi D, et al. Engineering of dendritic mesoporous silica nanoparticles for efficient delivery of waterinsoluble paclitaxel in cancer therapy. J Colloid Interface Sci. 2021; 593: 424-33. doi: 10.1016/j.jcis.2021.02.098.
Ma JZ, Sun HY, Zhang Y, Chen DW, Hu HY. Fabrication of epidermal growth factor imprinted and demethylcantharidin loaded dendritic mesoporous silica nanoparticle: An integrated drug vehicle for chemo-/antibody synergistic cancer therapy. J Drug Deliv Sci Technol. 2021; 62: 102387. doi:10.1016/j.jddst.2021.102387.
Xu Z, Keller E, Beltrán AM, Zheng K, Boccaccini AR. Cerium doped dendritic mesoporous bioactive glass nanoparticles with bioactivity and drug delivery capability. J Non Cryst Solids. 2023; 620: 122578. doi: 10.1016/j.jnoncrysol.2023.122578.
Pan Y, Zhou S, Li Y, Parshad B, Li W, Haag R. Novel dendritic polyglycerol-conjugated, mesoporous silica-based targeting nanocarriers for co-delivery of doxorubicin and tariquidar to overcome multidrug resistance in breast cancer stem cells. J Control Release. 2021; 330: 1106-17. doi: 10.1016/j.jconrel.2020.11.015.
Wu L, Lv R, Wang H, Gao S, Wang Y, Zhang S, et al. Facile fabrication of glutathione-responsive and photothermal nanocarriers with dendritic mesoporous silica nanoparticles for the controlled drug delivery. J Nanoparticle Res. 2022; 24(7): 128. doi: 10.1007/s11051-022-05498-0.
Ye M, Zhang W, Xu H, Xie P, Song L, Sun X, et al. Fe-doped biodegradable dendritic mesoporous silica nanoparticles for starvation therapy and photothermal-enhanced cascade catalysis in tumor therapy. J Colloid Interface Sci. 2025; 678: 378-92. doi: 10.1016/j.jcis.2024.08.172.
Song S, Li X, Ji Y, Lv R, Wu L, Wang H, et al. GSH/pH dual-responsive and HA-targeting nano-carriers for effective drug delivery and controlled release. J Drug Deliv Sci Technol. 2021; 62: 102327. doi: 10.1016/j.jddst.2021.102327.
Pota G, Andrés-Sanz D, Gallego M, Vitiello G, López-Gallego F, Costantini A, et al. Deciphering the immobilization of lipases on hydrophobic wrinkled silica nanoparticles. Int J Biol Macromol. 2024; 266: 131022. doi: 10.1016/j.ijbiomac.2024.131022.
Moon D-S, Lee J-K. Tunable synthesis of hierarchical mesoporous silica nanoparticles with radial wrinkle structure. Langmuir. 2012; 28(33): 12341-7. doi: 10.1021/la302145j.
Moon D-S, Lee J-K. Formation of wrinkled silica mesostructures based on the phase behavior of pseudoternary systems. Langmuir. 2014; 30(51): 15574-80. doi: 10.1021/la504207k.
Lo TNH, Park I. Unprecedented superoleophobicity achieved with fluorinated wrinkle mesoporous silica. Surfaces and Interfaces. 2024; 46: 104090. doi: 10.1016/j.surfin.2024.104090.
Flood-Garibay JA, Méndez-Rojas MA. Synthesis and characterization of magnetic wrinkled mesoporous silica nanocomposites containing Fe3O4 or CoFe2O4 nanoparticles for potential biomedical applications. Colloids Surfaces A Physicochem Eng Asp. 2021; 615: 126236. doi: 10.1016/j.colsurfa.2021.126236.
Flood-Garibay JA, Méndez-Rojas MA. A simple method for the synthesis of carboxymethylated wrinkled mesoporous silica nanoparticles and preparation of a WMS-curcumin conjugate. J Sol-Gel Sci Technol. 2022; 102(1): 288-95. doi: 10.1007/s10971-022-05747-7.
Kole K, Das S, Samanta A, Jana S. Parametric study and detailed kinetic understanding of CO2 adsorption over high-surface-area flowery silica nanomaterials. Ind Eng Chem Res. 2020; 59(49): 21393-402. doi: 10.1021/acs.iecr.0c04531.
Das S, Samanta A, Jana S. Light-driven synthesis of uniform dandelion-like mesoporous silica nanoflowers with tunable surface area for carbon dioxide uptake. Chem Eng J. 2019; 374: 1118-26. doi: 10.1016/j.cej.2019.05.114.
Yang H, Liao S, Huang C, Du L, Chen P, Huang P, et al. Facile one-pot approach to the synthesis of spherical mesoporous silica nanoflowers with hierarchical pore structure. Appl Surf Sci. 2014; 314: 7-14. doi: 10.1016/j.apsusc.2014.06.128.
Zhang H, Li Z, Xu P, Wu R, Jiao Z. A facile two step synthesis of novel chrysanthemum-like mesoporous silica nanoparticles for controlled pyrene release. Chem Commun. 2010; 46(36): 6783. doi: 10.1039/c0cc01673j.
Silmi N, Arsyad R, Benu DP, Nugroho FG, Khasannah WL, Iqbal M, et al. A morphological study of bicontinuous concentric lamellar silica synthesized at atmospheric pressure and its application as an internal micro-reflector in dyesensitized solar cells. Phys Chem Chem Phys.2023; 25(35): 23792–807. doi: 10.1039/D3CP02876C.
Febriyanti E, Silmi N, Suendo V, Mukti RR, Vivitasari PU, Adhika DR, et al. Thermodynamic Picture of Phase Segregation during the Formation of Bicontinuous Concentric Lamellar (bcl) Silica. Langmuir. 2022; 38(4): 1368–79. doi: 10.1021/acs.langmuir.1c02490.
Chiang Y-D, Lian H-Y, Leo S-Y, Wang S-G, Yamauchi Y, Wu KC-W. Controlling particle size and structural properties of mesoporous silica nanoparticles using the Taguchi method. J Phys Chem C. 2011; 115(27): 13158–65. doi: 10.1021/jp201017e.
Öztürk K, Kaplan M, Çalış S. Effects of nanoparticle size, shape, and zeta potential on drug delivery. Int J Pharm. 2024; 666: 124799. doi: 10.1016/j.ijpharm.2024.124799.