Developing a PET normal brain template using diffusion tensor imaging images: A proof of concept
Main Article Content
Abstract
Background: Registered Positron emission tomography (PET) brain images to the standard normal PET brain templates can be performed to diagnosis dementia by using a vendor software, in which the brain template is based on T1-Weighted (T1W) images. However, the imperfection of an overlap between PET images and the PET-T1W based brain template could be observed.
Objectives: This pilot study aimed to develop a new PET brain template and compare the accuracy of image registration between a conventional PET-T1W based brain template and our proposed PET-DTI based brain template.
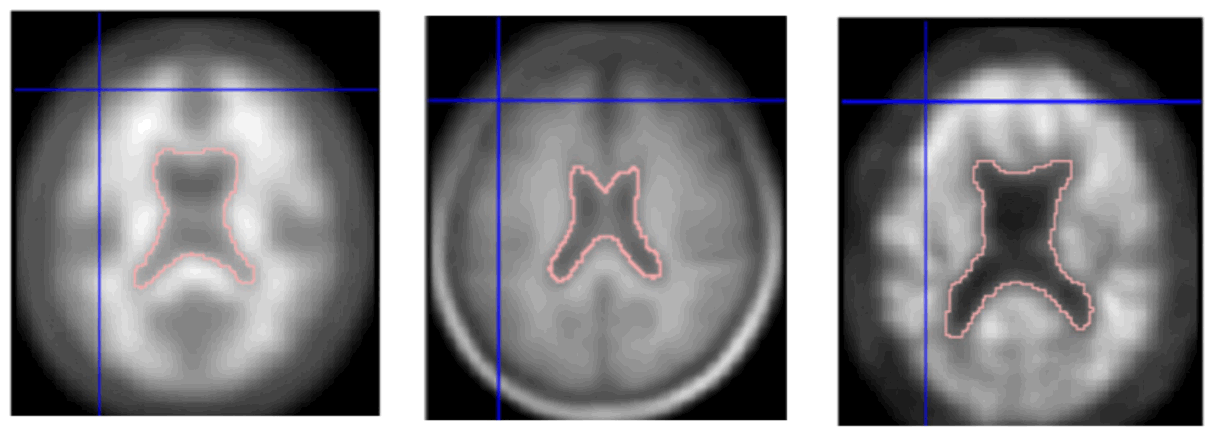
Materials and methods: The new PET-DTI based brain template was developed from twenty-four normal volunteers (age ranged 42-79 years old) who underwent 11C-Pittsburgh compound B PET scans and both T1W and diffusion tensor image (DTI) magnetic resonance imaging brain scans. The correction of Eddy-Current distortions and related artifact removing in DTI images were performed using the open-source FMRIB Software Library (FSL) to generate whole-brain probabilistic tractography maps (MRI-Probtract). MRI-Probtract map was then deformably registered and normalized to PET images, which were used for brain boundary guidance. The accuracy of image registration was assessed by applying the newly developed PET-DTI brain template to PET images of four mild cognitive impairment patients who underwent the same brain-scanning protocols. The accuracy of image registrations using the conventional PET-T1 and PET-DTI templates was evaluated qualitatively by three nuclear medicine physicians. Wilcoxon Signed Ranks test was used to compare registration scores of the two methods. Additionally, the dice similarly coefficient was obtained to quantitatively evaluate the accuracy of image registration.
Results: The registration scores of the PET images registered with the PET-DTI template were significantly higher than the PET-T1 template at p-value < 0.05. This result is consistent with the dice similarly coefficient where the value of PET-DTI template was higher.
Conclusion: Result of this pilot study showed that new PET-DTI brain template provides higher registration quality, suggesting the feasibility of using PET-DTI template in a clinical PET study of the brain.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Personal views expressed by the contributors in their articles are not necessarily those of the Journal of Associated Medical Sciences, Faculty of Associated Medical Sciences, Chiang Mai University.
References
Fletcher JW, Djulbegovic B, Soares HP, Siegel BA, Lowe VJ, Lyman GH, et al. Recommendations on the use of 18F-FDG PET in oncology. J Nucl Med. 2008; 49: 480-508. DOI: 10.2967/jnumed.10 7.047787.
Boellaard R, O’Doherty MJ, Weber WA, Mottaghy FM, Lonsdale MN, Stroobants SG, et al. FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2010; 37: 181-200. DOI: 10.1007/ s00259-009-1297-4.
Hofman MS, Hicks RJ. How we read oncologic FDG PET/CT. Cancer Imaging. 2016; 16: 1-4. DOI: https://doi.org/10.1186/s40644-016-0091-3.
Papathanassiou D, Bruna-Muraille C, Liehn JC, Nguyen TD, Curé H. Positron emission tomography in oncology: present and future of PET and PET/CT. Crit Rev Oncol Hematol. 2009; 72: 239-54. DOI: 10.1016/j.critrevonc.2008.10.006.
Burdette JH, Minoshima S, Vander Borght T, Tran DD, Kuhl DE. Alzheimer disease: improved visual interpretation of PET images by using three-dimensional stereotaxic surface projections. Radiology. 1996; 198: 837-43. DOI: 10.1148/radiology.198.3.8628880.
Xiong KL, Yang QW, Gong SG, Zhang WG. The role of positron emission tomography imaging of β-amyloid in patients with Alzheimer's disease. Nucl Med Commun. 2010; 31: 4-11. DOI: 10.1097/mnm.0b013e32833019f3.
Patterson JC, Lilien DL, Takalkar A, Pinkston JB. Early detection of brain pathology suggestive of early AD using objective evaluation of FDG-PET scans. Int J Alzheimer Dis. 2010; 2011: 946590. DOI: 10.4061/2011/946590D.
Daniela P, Orazio S, Alessandro P, Mariano NF, Leonardo I, Pasquale Anthony DR, et al. A survey of FDG and amyloid-PET imaging in dementia and GRADE analysis. Biomed Res Int. 2014; 785039. DOI: https://doi.org/10.1155/2014/785039.
Chotipanich C, Nivorn M, Kunawudhi A, Promteangtrong C, Boonkawin N, Jantarato A. Evaluation of imaging windows for Tau PET imaging using 18F-PI2620 in cognitively normal individuals, mild cognitive impairment, and Alzheimer’s disease patients. Mol Imaging. 2020; 19: 1-8. DOI: 10.1177/1536012120947582.
Jantarato A, Vachatimanont S, Boonkawin N, Yaset S, Kunawudhi A, Promteangtrong C, et al. The Evaluation of Tau Deposition with [18F] PI-2620 by Using a Semiquantitative Method in Cognitively Normal Subjects and Patients with Mild Cognitive Impairment and Alzheimer’s Disease. Mol Imaging. 2021; 2021: DOI: 10.1155/2021/6640054.
Jankovic J, Mazziotta JC, Newman NJ, Pomeroy SL. Diagnosis of Neurological Disease. Bradley and Daroff's Neurology in Clinical Practice. 8th ed. Philadelphia, PA: Elsevier. 2022.
Sungkarat W, Laothamatas J, Lumlertdacha B, Wacharapluesadee S, Hemachudha T.Dog’s Whole Brain Probabilistic Diffusion Tensor Imaging Tractography Normalization: A Solution for Brain Image Normalization Difficulty Problems. Proc. Intl. Soc. Mag. Reson. Med. 2012; 20: 3394.
Tritanon O, Laothamatas J, Wirojtananugoon C, Sungkarat W, Sukying C, Chansirikarnjana S. White Matter Involvement in Mild Cognitive Impairment and Mild Alzheimer’s Disease Using Whole-Brain Probabilistic Tractography Normalization. Proc. Intl. Soc. Mag. Reson. Med. 2012; 20: 979.
Kemppainen NM, Aalto S, Wilson IA, Någren K, Helin S, Brück A, et al. PET amyloid ligand [11C] PIB uptake is increased in mild cognitive impairment. Neurology. 2007; 68: 1603-6. DOI: 10.1212/01. wnl.0000260969.94695.56.
Rinne JO, Brooks DJ, Rossor MN, Fox NC, Bullock R, Klunk WE, et al. 11C-PiB PET assessment of change in fibrillar amyloid-β load in patients with Alzheimer's disease treated with bapineuzumab: a phase 2, double-blind, placebo-controlled, ascending-dose study. Lancet Neurol. 2010; 9: 363-72. DOI: 10.1016/S1474-4422(10)70043-0.
Zhang S, Smailagic N, Hyde C, Noel‐Storr AH, Takwoingi Y, McShane R, et al. 11C‐PIB‐PET for the early diagnosis of Alzheimer’s disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2014; 7: DOI:10.1002/14651858.CD0 10386.pub2.
Chotipanich C, Kongthai S, Kunawudhi A, Promteangtrong C, Jantarato A. 18F-THK 5351 and 11C-PiB PET of the Thai normal brain template. Asia Ocean J Nucl Med Biol. 2021; 9: 21. DOI: 10.22038/AOJNMB.2020.49623.1338.
Woolrich MW, Jbabdi S, Patenaude B, Chappell M, Makni S, Behrens T, Beckmann C, et al. Bayesian analysis of neuroimaging data in FSL. Neuroimage. 2009; 45: S173-86. DOI: 10.1016/j.neuroimage. 2008.10.055.
Jbabdi S, Sotiropoulos SN, Savio AM, Graña M, Behrens TE. Model‐based analysis of multishell diffusion MR data for tractography: How to get over fitting problems. Magn Reson Med. 2012; 68(6): 1846–1855. DOI:10.1002/mrm.24204.
Rao NP, Jeelani H, Achalia R, Achalia G, Jacob A, dawn Bharath R, et al. Population differences in brain morphology: need for population specific brain template. Psychiatry Res. 2017; 265: 1-8.DOI:10.1016/j.pscychresns.2017.03.018.